Tailoring the local environment around metal ions: a solution chemical and structural study of some multidentate tripodal ligands†
Abstract
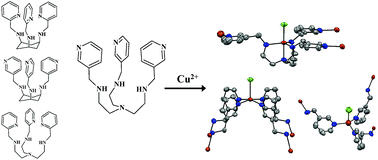
Manganese(II), copper(II) and zinc(II) complexes of four polydentate tripodal ligands (tachpyr (N,N′,N′′-tris(2-pyridylmethyl)-cis,cis-1,3,5-triaminocyclohexane), trenpyr (tris[2-(2-pyridylmethyl)aminoethyl]amine, tach3pyr (N,N′,N′′-tris(3-pyridylmethyl)-cis,cis-1,3,5-triaminocyclohexane) and tren3pyr (tris[2-(2-pyridylmethyl)aminoethyl]amine)) were characterized in both solution and solid states. A combined evaluation of potentiometric, UV-VIS, NMR and EPR data allowed the conclusion of both thermodynamic and structural information about the complexes formed in solution. The four tailored polydentate tripodal ligands studied here exhibit a high thermodynamic stability, and a variety of coordination environments/geometries for the studied transition metal ions. Our data indicate that tachpyr is a more efficient zinc(II) chelator and a similar copper(II) chelator compared to trenpyr. Considering the higher number of N-donors and conformational flexibility of trenpyr, as well as the energy demanding switch to the triaxial conformation required for metal ion binding of tachpyr, the above observation is surprising and is very likely due to the encapsulating effect of the more rigid tachpyr skeleton. This relative binding preference of tachpyr for zinc(II) may be related to the observation that zinc(II) is one of the principal metals targeted by tachpyr in cells. In contrast, trenpyr is a considerably more efficient manganese(II) chelator, since it acts as a heptadentate ligand in the aqueous Mn(trenpyr) complex. The crystal structures of copper(II) and zinc(II) complexes of tachpyr indicated important differences in the ligand conformation, induced by the position of counter ions, as compared to earlier reports. The closely related new ligands, tach3pyr and tren3pyr, have been designed to form oligonuclear complexes. Indeed, we obtained a three dimensional polymer with a copper(II)/tren3pyr ratio of 11/6. Within this metal–organic framework, three distinctly different copper geometries can be identified: square pyramidal, trigonal bipyramidal and tetrahedral. Two square pyramidal and four trigonal bipyramidal copper centres create a hexanuclear subunit with a large inside cavity. These moieties are linked by tetrahedral copper(II) centres, constructing the three-dimensional polymer structure. The formation of such polynuclear complexes was not detected in solution. Both tach3pyr and tren3pyr form only mononuclear complexes with square pyramidal and trigonal bipyramidal geometries, respectively.


 Please wait while we load your content...
Please wait while we load your content...