Analysis of crystallographic structures of Ni(ii) complexes of α-amino acid Schiff bases: elucidation of the substituent effect on stereochemical preferences†
Abstract
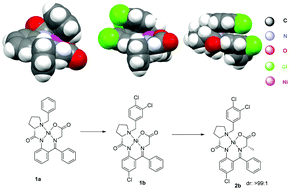
In this study, we performed critical analysis of 13 crystallographic structures of various Ni(II) complexes of amino acid Schiff bases. The major finding of this work is the significance of a parallel displaced type of aromatic interactions between o-amino-benzophenone and Pro N-benzyl rings. The quality of these aromatic interactions was shown to control the steric environment around the amino acid side-chain, rendering variously substituted Ni(II) complexes of different thermodynamic stabilities. The discovered structural trend holds true for aliphatic, aromatic and sterically bulky amino acids and can be used for the rational design of new and more efficient chiral ligands for general asymmetric synthesis of tailor-made α-amino acids.


 Please wait while we load your content...
Please wait while we load your content...