Synthesis and photocatalytic activity of K2CaNaNb3O10, a new Ruddlesden–Popper phase layered perovskite†
Abstract
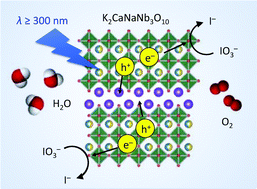
A new three-layer perovskite oxide with the Ruddlesden–Popper (R–P) phase, K2CaNaNb3O10, and its protonated form were synthesised and their photocatalytic performance was compared to that of KCa2Nb3O10 or the protonated form with the Dion–Jacobson (D–J) structure in terms of H2 and O2 evolution. K2CaNaNb3O10 exhibited a higher activity for O2 evolution than KCa2Nb3O10 when IO3− was used as an electron acceptor. However, protonated KCa2Nb3O10 worked more efficiently than protonated K2CaNaNb3O10 when Fe3+ was used as an electron acceptor. In both cases, it is likely that the stronger affinity of water with the interlayer contributed to higher performance. The activity of the D–J material for H2 evolution was much lower when 1-propanol was used as an electron donor than when methanol was used. In contrast, the R–P phase exhibited a similar activity regardless of the electron donor. These results indicate that the interlayer space acts as an oxidation site, so that better access of the electron donor to the interlayer is an important factor that enhances photocatalytic activity.
- This article is part of the themed collection: The Role of Inorganic Materials in Renewable Energy Applications


 Please wait while we load your content...
Please wait while we load your content...