Surface chemistry of pure tetragonal ZrO2 and gas-phase dependence of the tetragonal-to-monoclinic ZrO2 transformation†
Abstract
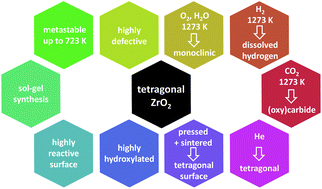
The surface chemical properties of undoped tetragonal ZrO2 and the gas-phase dependence of the tetragonal-to-monoclinic transformation are studied using a tetragonal ZrO2 polymorph synthesized via a sol–gel method from an alkoxide precursor. The obtained phase-pure tetragonal ZrO2 is defective and strongly hydroxylated with pronounced Lewis acidic and Brønsted basic surface sites. Combined in situ FT-infrared and electrochemical impedance measurements reveal effective blocking of coordinatively unsaturated sites by both CO and CO2, as well as low conductivity. The transformation into monoclinic ZrO2 is suppressed up to temperatures of ∼723 K independent of the gas phase composition, in contrast to at higher temperatures. In inert atmospheres, the persisting structural defectivity leads to a high stability of tetragonal ZrO2, even after a heating–cooling cycle up to 1273 K. Treatments in CO2 and H2 increase the amount of monoclinic ZrO2 upon cooling (>85 wt%) and the associated formation of either Zr-surface-(oxy-)carbide or dissolved hydrogen. The transformation is strongly affected by the sintering/pressing history of the sample, due to significant agglomeration of small crystals on the surface of sintered pellets. Two factors dominate the properties of tetragonal ZrO2: defect chemistry and hydroxylation degree. In particular, moist conditions promote the phase transformation, although at significantly higher temperatures as previously reported for doped tetragonal ZrO2.


 Please wait while we load your content...
Please wait while we load your content...