Divergent reactivity of a new dinuclear xanthene-bridged bis(iminopyridine) di-nickel complex with alkynes†
Abstract
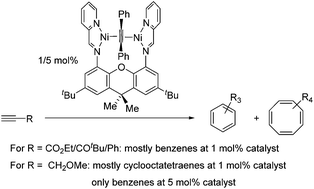
The reaction of a dinucleating bis(iminopyridine) ligand L bearing a xanthene linker (L = N,N′-(2,7-di-tert-butyl-9,9-dimethyl-9H-xanthene-4,5-diyl)bis(1-(pyridin-2-yl)methanimine)) with Ni2(COD)2(DPA) (COD = cyclooctadiene, DPA = diphenylacetylene) leads to the formation of a new dinuclear complex Ni2(L)(DPA). Ni2(L)(DPA) can also be obtained in a one-pot reaction involving Ni(COD)2, DPA and L. The X-ray structure of Ni2(L)(DPA) reveals two square-planar Ni centers bridged by a DPA ligand. DFT calculations suggest that this species features NiI centers antiferromagnetically coupled to each other and their iminopyridine ligand radicals. Treatment of Ni2(L)(DPA) with one equivalent of ethyl propiolate (HCCCO2Et) forms the Ni2(L)(HCCCO2Et) complex. Addition of the second equivalent of ethyl propiolate leads to the observation of cyclotrimerised products by 1H NMR spectroscopy. Carrying out the reaction under catalytic conditions (1 mol% of Ni2(L)(DPA), 24 h, room temperature) transforms 89% of the substrate, forming primarily benzene products (triethyl benzene-1,2,4-tricarboxylate and triethyl benzene-1,3,5-tricarboxylate) in 68% yield, in a ca. 5 : 1 relative ratio. Increasing catalyst loading to 5 mol% leads to the full conversion of ethyl propiolate to benzene products; no cyclotetramerisation products were observed. In contrast, the reaction is significantly more sluggish with methyl propargyl ether. Using 1 mol% of the catalyst, only 25% conversion of methyl propargyl ether was observed within 24 h at room temperature. Furthermore, methyl propargyl ether demonstrates the formation of cyclooctatetraenes in significant amounts at a low catalyst concentration, whereas a higher catalyst concentration (5 mol%) leads to benzene products exclusively. Density functional theory was used to provide insight into the reaction mechanism, including structures of putative dinuclear metallocyclopentadiene and metallocycloheptatriene intermediates.
- This article is part of the themed collection: Multimetallic complexes: synthesis and applications


 Please wait while we load your content...
Please wait while we load your content...