Structure and catalytic activity of a newly proposed titanium species in a Ti-YNU-1 zeolite: a density functional theory study†
Abstract
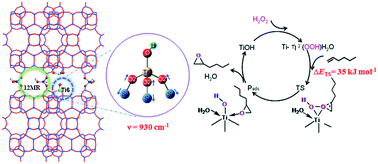
Density functional theory was applied to investigate the structure of the framework titanium (Ti) species in the Ti-YNU-1 zeolite, and to evaluate its catalytic activity for 1-hexene epoxidation with H2O2 as the oxidant. The effects of solvent coordination on the stability and reactivity of Ti-hydroperoxo intermediates were studied. All calculations were performed at the theoretical level of B3LYP/6-31G(d,p) based on cluster models. Results suggested that a defective Ti-hydroxyl species [Ti(OSi)3OH] at the pillar (T6) site between the interlayers in the Ti-YNU-1 zeolite was responsible for the Ti-specific vibrational frequency observed at 930 cm−1, and was in good agreement with the experimental infrared spectra obtained for the Ti-rich Ti-YNU-1 zeolite. The stabilities of the possible Ti-hydroperoxo intermediates decreased in the order Ti-η2(OOH)–H2O > Ti-η2(OOH)–CH3CN > Ti-η2(OOH) as well as Ti-η2(OOH)–H2O > Ti-η1(OOH). The activation barriers of 1-hexene epoxidation over Ti6-η2(OOH), Ti6-η2(OOH)-H2O, and Ti6-η2(OOH)-CH3CN were 33.2, 35.0, and 43.9 kJ mol−1, respectively. The Ti6-η1(OOH) species possessed lower stability and had structural changes in the transition state for 1-hexene epoxidation, leading to the same transition-state structure as that of the bidentate Ti-η2(OOH)–H2O species. For comparison, the reactivity of the perfect Ti(OSi)4 species at the T1 site in the Ti-MWW zeolite was also investigated. The transition state of Ti1-η1(OOH) was obtained with an activation barrier of 107.2 kJ mol−1, which was much higher than that of Ti1-η2(OOH)–H2O. This result revealed that the intramolecular hydrogen bonding in the monodenate five-membered ring structure did not enhance the stability and reactivity of Ti-hydroperoxo intermediates. These results also indicated that Ti-YNU-1 was more active than Ti-MWW, which was in good agreement with experimental results.

 Please wait while we load your content...
Please wait while we load your content...