Mechanistic study of the role of Au, Pd and Au–Pd in the surface reactions of ethanol over TiO2 in the dark and under photo-excitation†
Abstract
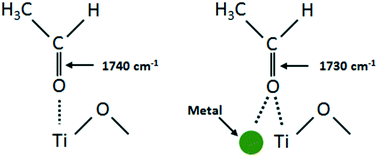
In situ infrared spectroscopy (FTIR) and catalytic reactions are employed to explore the photo-oxidation and photo-reforming of ethanol over TiO2 and M/TiO2 (M = Au, Pd and Au–Pd) catalysts. The effect of metal loadings (in mono- and bimetallic systems), the presence and absence of gas phase oxygen and the role of water are investigated. TEM images of the Au–Pd/TiO2 sample indicate that the mean Au particle size is 3.6 nm while that of Pd is 1.4 nm. Reaction intermediates on the surface during photo-oxidation of ethanol include carbon monoxide, acetaldehyde, acetate, formate and carbonate/bicarbonate species. The extent of formation and stability of adsorbed acetaldehyde, generated by dehydrogenation of ethanol, are in the order Au–Pd/TiO2 > Pd/TiO2 > Au/TiO2 > TiO2. Carboxylates predominantly appeared on all catalysts, while carbon monoxide (under inert flow) and carbonates/bicarbonates (under an O2 environment) are specifically produced on catalysts containing higher loading of Pd in both mono- and bimetallic systems. During photo-catalytic hydrogen production in the presence of ethanol/water at a light flux of 4–5 mW cm−2 (excitation energy = 3.44 eV), a maximum hydrogen production rate of 2.0 × 10−4 mol min−1 gcatalyst−1 is obtained with the Au–Pd/TiO2 catalyst (higher than those on its monometallic counterparts). The presence of Au in the bimetallic catalysts kept Pd in its metallic state as evidenced from UV-vis and X-ray photoelectron spectroscopy studies due to the enhanced carrier dynamics, facilitating charge transfer from Au to Pd, generating more hydrogen. The turnover frequency (TOF) per metal atom increases in the case of Au–Pd/TiO2 when compared to those of the monometallic catalysts (Au/TiO2 and Pd/TiO2 at similar loadings). In addition to hydrogen, during the initial period of the photo-reaction, acetaldehyde (in both the gas and dissolved phases) and methane (in the gas phase) are produced, suggesting that ethanol in aqueous solution reacted through dehydrogenation as well as C–C bond cleavage pathways. As the reaction proceeds, carbon dioxide is released following the same trend as that of methane. Results from this study are discussed in order to provide a comprehensive reaction network needed to understand the key parameters behind the photo-catalytic reaction on metal/semiconductor materials.
- This article is part of the themed collection: Catalytic reactivity of surfaces: in recognition of François Gault


 Please wait while we load your content...
Please wait while we load your content...