Activation and deactivation of Ag/CeO2 during soot oxidation: influences of interfacial ceria reduction†
Abstract
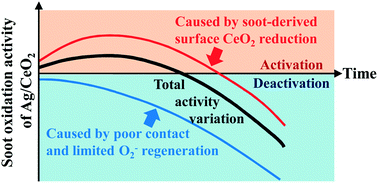
Different Ag/CeO2 catalysts with uniform structures and diverse surface oxygen vacancy (VO-s) contents were synthesized via a modified solution combustion method. Based on the results of Raman spectroscopy, H2-TPR and isothermal soot oxidation, it was suggested that even with gaseous O2 participating in the reaction, ceria surface sites around the catalyst–soot contact points can still be reduced by soot gradually. This interfacial reduction led to the generation of extra VO-s without changing the oxygen vacancies in the bulk of CeO2. Notably, this VO-s dynamic change and the insufficient O2− regeneration/delivery further resulted in the activation and deactivation of Ag/CeO2 during reaction with soot. This means that both the existence of ceria VO-s and its dynamic change during reaction may have a crucial influence on catalyst behavior. By further exploring these activity variations, the VO-s content, catalyst–soot contact condition and extra oxygen supplier were proved to be the three most important structural factors behind the soot oxidation activity of all the ceria-based catalysts. In this sense, Ag/CeO2 catalysts with appropriate morphology and moderate initial VO-s content may exhibit good activity for soot oxidation.


 Please wait while we load your content...
Please wait while we load your content...