Direct cyanomethylation of aliphatic and aromatic hydrocarbons with acetonitrile over a metal loaded titanium oxide photocatalyst†
Abstract
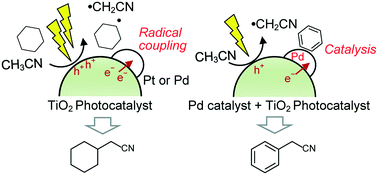
A platinum-loaded TiO2 (Pt/TiO2) photocatalyst promoted cyanomethylation of aliphatic hydrocarbons, namely cyclohexane and cyclohexene, with acetonitrile, where the photogenerated hole oxidatively dissociates the C–H bond of both the acetonitrile and the aliphatic hydrocarbons to form each corresponding radical species before their radical cross-coupling. The Pt/TiO2 photocatalyst was more active than the Pd/TiO2 photocatalyst in these reactions. In contrast, the cyanomethylation of benzene was promoted by the Pd/TiO2 photocatalyst or a physical mixture of the Pt/TiO2 photocatalyst and a Pd catalyst supported by Al2O3, while it was hardly promoted by the Pt/TiO2 photocatalyst alone. The temperature dependence of the reaction rate proved that the Pd nanoparticles on the TiO2 photocatalyst thermally function as a metal catalyst. These results clearly suggest that the Pd metal catalyst is necessary for the cyanomethylation of benzene. However, in the cyanomethylation of aliphatic hydrocarbons, the catalytic effect of the metal particles was not observed, meaning that the radical coupling takes place without the metal catalysis. Thus, it is concluded that in the case of the benzene cyanomethylation the Pd nanoparticles play dual roles, as a catalyst to catalyse the substitution reaction of benzene with the cyanomethyl radical, and as an electron receiver to reduce the recombination of the photoexcited electrons and holes in the TiO2 photocatalyst, although they could not contribute as a catalyst to the cyanomethylation of aliphatic hydrocarbons.


 Please wait while we load your content...
Please wait while we load your content...