Elucidating the reaction pathway for ethene and propene formation in the methanol-to-hydrocarbons reaction over high silica H-Beta†
Abstract
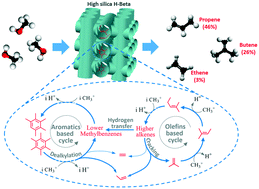
The methanol-to-hydrocarbons reaction over high silica H-Beta was investigated in a fixed bed reactor. It was found to be a promising catalyst for methanol conversion, exhibiting a high selectivity to propene and low selectivity to ethene, and a high propene/ethene ratio of 11–28. Meanwhile, a long lifetime was obtained under the operating conditions (reaction temperature = 450 °C, PMeOH = 17 kPa, and WHSV = 2 h−1). The underlying reaction pathway was studied by multiple approaches (steady-state, transient, and co-feed) to elucidate the mechanism for ethene and propene formation. The olefins-based cycle was proved to be an important pathway for the formation of C3+ olefins over the H-Beta through fast methylation and cracking of higher olefins. In addition, the results demonstrated that ethene could be a product of the olefins-based cycle, even though in a much lesser content than propene. On the other hand, due to the large interspace of H-Beta, the lower methylbenzenes are prone to be methylated when introduced to or formed in the pores, resulting in the domination of pentamethylbenzene and hexamethylbenzene in the species retained in the catalyst, which works in the aromatics-based cycle. Thus, the olefins-based cycle as well as aromatics-based cycle involving the higher methylbenzenes contributes to the high selectivity for propene while the ethene formation was limited in both cycles. This work provides new insight into the dual-cycle concept and the design of more efficient catalysts for methanol conversion.


 Please wait while we load your content...
Please wait while we load your content...