Mechanistic insights into the remarkable catalytic activity of nanosized Co@C composites for hydrogen desorption from the LiBH4–2LiNH2 system†
Abstract
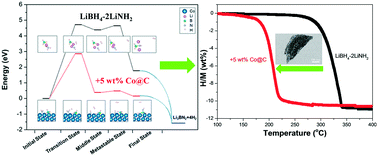
In this work, we demonstrate a first attempt at understanding the catalytic mechanism of nanosized Co in reducing the dehydrogenation temperature of the Li–B–N–H hydrogen storage system by experimental observation and theoretical calculation. A nanosized Co@C composite (Co particles <10 nm) is successfully synthesized by casting a furfuryl alcohol-filled, Co-based metal–organic framework, MOF-74(Co), at 700 °C. Adding small quantities of the prepared nanosized Co@C composite significantly reduces the dehydrogenation temperature of the LiBH4–2LiNH2 system. The 5 wt% Co@C-containing sample releases approximately 10.0 wt% hydrogen at 130–230 °C with a peak temperature of 210 °C, which is reduced by 125 °C from that of the pristine sample. During hydrogen desorption, nanosized Co remains in the metallic state and only works as a catalyst to reduce the kinetic barriers of hydrogen release from the LiBH4–2LiNH2 system. Ab initio calculations reveal that the presence of a Co catalyst induces a redistribution of charge, which not only weakens the chemical H–B bonding but also enhances the electrostatic interactions between Hδ+ in the NH2 groups and Hδ− in the BH4 groups, consequently reducing the energy barriers for the formation of H2 molecules. This explains the low-temperature dehydrogenation behaviour of the Co-catalysed Li–B–N–H systems.


 Please wait while we load your content...
Please wait while we load your content...