Design strategies of surface basicity for NO oxidation over a novel Sn–Co–O catalyst in the presence of H2O†
Abstract
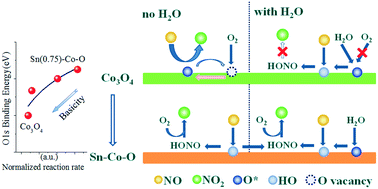
The influence of H2O on NO oxidation has attracted much attention recently. Herein, a series of novel SnO2–CoOx catalysts were designed and prepared to modify the surface basicity of Co3O4 catalyst. CO2-TPD results suggested that the basicity of Co3O4 was weakened by Sn doping, and that the relative ratio of very strong basic sites had decreased on SnO2–CoOx. In the presence of H2O, the catalyst with a Sn : Co molar ratio of 3 : 1 (denoted as Sn(0.75)–Co–O) exhibited excellent NO oxidation activity in the temperature range 250–300 °C. NO-TPD showed that additional NO adsorption sites (with H2O) appeared after Sn doping. In both the absence and presence of H2O, the amount of adsorbed NOx was larger compared to Co3O4. XPS spectra showed that the ratio of chemisorbed oxygen increased, which contributed to the enhanced redox properties of the Sn(0.75)–Co–O catalyst. Meanwhile, the NO oxidation reaction rate, normalized to surface Co concentration, increased with increasing lattice oxygen binding energy, which demonstrated that the NO oxidation reaction rate (with H2O) increased as basicity decreased. Moreover, the enlarged BET specific surface area was favourable for NO oxidation. In situ DRIFTS studies proposed that nitrite/HONO species were active intermediates in NO oxidation over the Sn(0.75)–Co–O catalyst. A novel mechanism was proposed, including suppression of O2 adsorption by H2O and new OH sites providing additional sites for NO adsorption. The improved H2O resistance is essential for the application of these environmentally friendly catalysts to NO oxidation.


 Please wait while we load your content...
Please wait while we load your content...