Hypervalency, secondary bonding and hydrogen bonding: siblings under the skin
Abstract
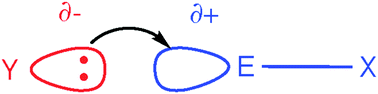
In hypervalent bonding (HVB), secondary bonding (SB) and hydrogen bonding (HB) a nucleophilic and an electrophilic partner form a new bond that is based on a similar bonding pattern across the whole series of interactions. The electrostatic contribution is reflected in the ‘σ hole’ model in which a positive patch on E attracts the nucleophilic component. The nucleophile, Y, possesses a corresponding negative patch, resulting in a linear structure Y⋯E–X having one strong E–X bond and one weaker, longer Y⋯E interaction; this is considered as a SB interaction between Y and E. The covalent component, more important in the stronger interactions, HVB and strong HB, involves charge transfer between the lone pair (n) of Y, and the σ* orbital of E–X as emphasized in the ‘n→σ*’ bonding model. For example, charge transfer from I− to I2 gives rise to the linear, symmetrical [I–I–I]− anion. We now have two short (2.95 Å) bonds of equal strength corresponding to true HVB. In HB the central element, E, is H, and we can have strong or weak hydrogen bonding. On the HVB/HB analogy, a strong symmetrical HB, as in [F–H–F]−, can be considered as containing hypervalent hydrogen. In the weak HB case, we have a lesser degree of interaction, leading to normal hydrogen bonds of type Y⋯H–X analogous to secondary bonding. Within both the HB and HVB series, strong and weak types form a smooth continuum with no sharp break in properties. HVB was once considered to involve the expansion of the octet to 10, 12 or even higher valence electron counts. Whether the σ hole or n→σ* model applies, any octet expansion is now seen as largely formal, however, because the central element essentially retains its eight valence electrons. Thus a range of interactions can be placed in one big tent, related by a combination of σ hole and n→σ* bonding contributions with retention of the octet by the central element, E.
- This article is part of the themed collection: Primer


 Please wait while we load your content...
Please wait while we load your content...