Synergism and aggregation behaviour in an aqueous binary mixture of cationic–zwitterionic surfactants: physico-chemical characterization with molecular simulation approach
Abstract
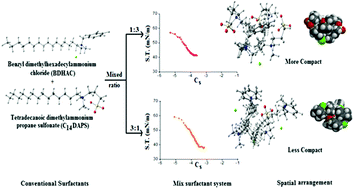
Aqueous interactions between a cationic surfactant benzyl dimethylhexadecylammonium chloride (BDHAC) and alkyldimethylammoniopropane sulfonates (CnDAPS) based three zwitterionic surfactants n = 10, 12, and 14 (abbreviated as C10DAPS, C12DAPS and C14DAPS, respectively) were studied using tensiometry, and fluorescence spectrophotometry techniques. The critical micelle concentration degree of synergism and various other parameters such as interaction parameter (β), activity coefficients (fm) and interfacial parameters such as surface pressure (πCMC), packing parameter (P), surface excess concentration (Γmax), surface tension at CMC (γCMC), and minimum area per molecule (Amin) were evaluated using the Regular Solution Theory (RST) of mixed systems. The results indicate a strong dependency on the mixed system and their composition. For the quantitative prediction, the molecular architecture of the surfactants in mixed systems and their synergistic interactions were investigated by computational simulation using Spartan’14 V1.1.8. The structural optimization results obtained were found to be in good agreement with the estimations made using RST. The reduction in surface tension indicates a certain efficiency in mixed micelle formation owing to electrostatic attraction between the cationic and zwitterionic surfactants. In addition, the binary surfactant systems evaluated by Maeda's approach infer the mixed micelles are thermodynamically stable. The aggregation number (Nagg) appeared to be larger at the composition point where the efficiency of mixed micelle formation is greatest. The strength of the interaction between BDHAC and CnDAPS followed the order: C14DAPS > C12DAPS > C10DAPS indicating a greater synergism at 0.25 molar ratio of zwitterionic surfactants to cationic surfactants in the aqueous solution at 303.15 K.


 Please wait while we load your content...
Please wait while we load your content...