Ostwald ripening versus single atom trapping: towards understanding platinum particle sintering†
Abstract
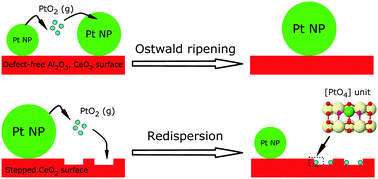
Ostwald ripening is a leading cause of the degradation of platinum group catalysts at high temperature in an oxidizing atmosphere. Recent experiments suggested that volatile species can be trapped on ceria, forming atomically dispersed active catalytic sites instead of large nanoparticles. Here we present a comparative density functional theory study of the interaction of PtO2(g), the most likely volatile species responsible for the process of Ostwald ripening, with various surfaces. Defect-free CeO2(111) and Al2O3(100) surfaces have a very small binding energy towards PtO2(g) compared to the platinum surface, indicative of particle growth. However, the stepped edge of the CeO2(111) surface effectively traps the mobile species, generating atomically dispersed catalysts. Such trapped single-atom platinum-on-ceria catalysts are predicted to have a square-planar [PtO4] structure, with the platinum atom strongly binding to the surface, preventing platinum atoms from aggregating into larger nanoparticles. These results provide an atomic insight into the single atom trapping and suggest a route for the development of sinter-resistant catalysts.


 Please wait while we load your content...
Please wait while we load your content...