Spectroscopic identification of the chemical interplay between defects and dopants in Al-doped ZnO
Abstract
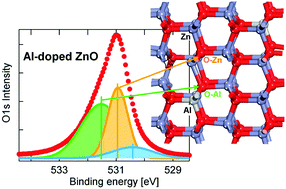
The conduction and optoelectronic properties of transparent conductive oxides can be largely modified by intentional inclusion of dopants over a very large range of concentrations. However, the simultaneous presence of structural defects results in an unpredictable complexity that prevents a clear identification of chemical and structural properties of the final samples. By exploiting the unique chemical sensitivity of Hard X-ray Photoelectron Spectra and Near Edge X-ray Absorption Fine Structure in combination with Density Functional Theory, we determine the contribution to the spectroscopic response of defects in Al-doped ZnO films. Satellite peaks in O1s and modifications at the O K-edge allow the determination of the presence of H embedded in ZnO and the very low concentration of Zn vacancies and O interstitials in undoped ZnO. Contributions coming from substitutional and (above the solubility limit) interstitial Al atoms have been clearly identified and have been related to changes in the oxide stoichiometry and increased oxygen coordination, together with small lattice distortions. In this way defects and doping in oxide films can be controlled, in order to tune their properties and improve their performances.


 Please wait while we load your content...
Please wait while we load your content...