An analysis of the explosion limits of hydrogen/oxygen mixtures with nonlinear chain reactions
Abstract
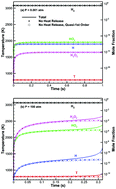
The ignition boundary of hydrogen/oxygen mixtures is a Z-shaped curve in the pressure–temperature space, demonstrating the existence of three explosion limits. In this study, a general analysis governing all the three explosion limits in an isothermal environment is performed by considering both linear chain reactions (reactant-radical reactions) and nonlinear chain reactions (radical–radical reactions), in addition to the zeroth-order reactant–reactant reactions. For the nonlinear reactions, it is further shown that the linear–nonlinear coupling has the major influence, while the effect of nonlinear–nonlinear coupling is negligible. Phenomenologically, at low pressures, the competition between linear branching and linear termination as well as wall destruction determines the first and second explosion limits, while the nonlinear chain reactions are unimportant because of the small radical concentrations under these conditions. However, at higher pressures, both linear and nonlinear chain reactions are needed to accurately describe the third limit, which would be underpredicted by considering the linear chain reactions alone. For intermediate and high pressures, the dominant species are HO2 and H2O2, respectively. Mechanistically, the concentration of HO2 becomes higher at higher pressures due to the three-body recombination reaction, H + O2 + M → HO2 + M, such that the radical–radical reactions involving HO2 become important, while the reaction HO2 + HO2 → H2O2 + O2 renders HO2 nonessential at the third limit, with the H2O2 radical generated by the nonlinear chain reactions becoming the controlling species.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...