Unified elucidation of the entropy-driven and -opposed hydrophobic effects†
Abstract
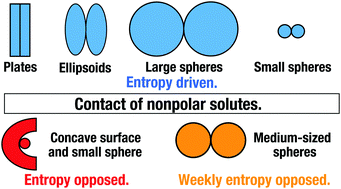
The association of nonpolar solutes is generally believed to be entropy driven, which has been shown to be true for the contact of small molecules, ellipsoids, and plates. However, it has been reported with surprise that a model cavity–ligand binding is entropy opposed. How can these apparently conflicting behaviors be elucidated? Here, we calculate the potential of mean force between hard-sphere solutes with various diameters in water and its entropic and enthalpic components using a statistical–mechanical theory for molecular liquids. It is shown that there is a very wide region where both of the two components are negative and large with the entropy–enthalpy compensation. Even for spheres, their contact is weakly entropy opposed when they are medium-sized. The entropic component (EC) is decomposed into physically insightful constituents with the aid of our morphometric approach. They provide us with useful information on the signs and magnitudes of contributions from the structural difference between the water near a single solute surface and that within the space confined between two solute surfaces and from the total volume available for the translational displacement of water molecules in the system. The decomposition enables us to identify the essential factors for discussing the EC: the hydrogen-bonding properties and density structure of the water within the confined space and the degree of water crowding in the bulk. These are largely dependent on the geometric characteristics of the solute pair such as solute shape, size, and intersolute distance. Both of the entropy-driven and -opposed hydrophobic effects can be explained within the same theoretical framework.
- This article is part of the themed collection: 2017 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...