Fabrication of photoluminescent nc-Si:SiO2 thin films prepared by PLD
Abstract
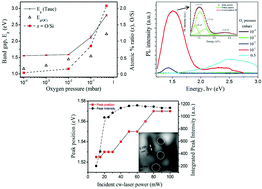
In the present report, the structural, compositional, morphological, and photoluminescence properties of nanostructured non-stoichiometric silicon oxide (nc-Si:SiO2 or SiOx) thin films fabricated by pulsed-laser ablation of silicon in the presence of oxygen pressure, from 10−4 to 0.5 mbar, are presented. X-ray diffraction spectra and Raman spectra confirmed the formation of nanocrystalline Si within the films while electron diffraction X-ray spectroscopy confirmed the increase in oxygen content with increasing O2 pressure. Scanning electron microscopy images of the SiOx films showed spreading of the micron-sized clusters on the otherwise uniform background, while Raman maps confirm the presence of nanocrystalline Si in these clusters embedded in a uniform matrix comprising oxidized amorphous silicon. A systematic blue shift in the band gap energy from 1.55 to 2.80 eV was observed with increasing O2 pressure in the SiOx films due to a shift in the stoichiometry of the films from x = 0.03 to 2.14 respectively. The films with higher oxygen content exhibited broad and intense PL emissions with multiple peaks originating from quantum confined (QC) Si nanocrystals as well as oxygen defects like NBOH and VO centers. The variation in PL intensity as a function of excitation intensity displays an initial linear increase followed by saturation, a characteristic feature of emissions from QC nc-Si.


 Please wait while we load your content...
Please wait while we load your content...