The effect of defects on the catalytic activity of single Au atom supported carbon nanotubes and reaction mechanism for CO oxidation†
Abstract
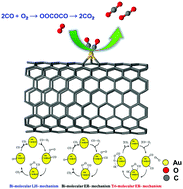
The mechanism of CO oxidation by O2 on a single Au atom supported on pristine, mono atom vacancy (m), di atom vacancy (di) and the Stone Wales defect (SW) on single walled carbon nanotube (SWCNT) surface is systematically investigated theoretically using density functional theory. We determine that single Au atoms can be trapped effectively by the defects on SWCNTs. The defects on SWCNTs can enhance both the binding strength and catalytic activity of the supported single Au atom. Fundamental aspects such as adsorption energy and charge transfer are elucidated to analyze the adsorption properties of CO and O2 and co-adsorption of CO and O2 molecules. It is found that CO binds stronger than O2 on Au supported SWCNT. We clearly demonstrate that the defected SWCNT surface promotes electron transfer from the supported single Au atom to O2 molecules. On the other hand, this effect is weaker for pristine SWCNTs. It is observed that the high density of spin-polarized states are localized in the region of the Fermi level due to the strong interactions between Au (5d orbital) and the adjacent carbon (2p orbital) atoms, which influence the catalytic performance. In addition, we elucidate both the Langmuir–Hinshelwood (LH) and Eley–Rideal (ER) mechanisms of CO oxidation by O2. For the LH pathway, the barriers of the rate-limiting step are calculated to be 0.02 eV and 0.05 eV for Au/m-SWCNT and Au/di-SWCNT, respectively. To regenerate the active sites, an ER-like reaction occurs to form a second CO2 molecule. The ER pathway is observed on Au/m-SWCNT, Au/SW-SWCNT and Au/SWCNT in which the Au/m-SWCNT has a smaller barrier. The comparison with a previous study (Lu et al., J. Phys. Chem. C, 2009, 113, 20156–20160.) indicates that the curvature effect of SWCNTs is important for the catalytic property of the supported single Au. Overall, Au/m-SWCNT is identified as the most active catalyst for CO oxidation compared to pristine SWCNT, SW-SWCNT and di-SWCNT. Our findings give a clear description on the relationship between the defects in the support and the catalytic properties of Au and open a new avenue to develop carbon nanomaterial-based single atom catalysts for application in environmental and energy related fields.


 Please wait while we load your content...
Please wait while we load your content...