Comparison of classical reaction paths and tunneling paths studied with the semiclassical instanton theory†
Abstract
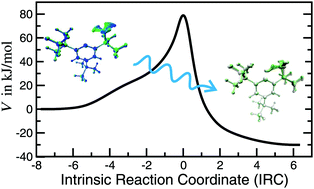
Atom tunneling in the hydrogen atom transfer reaction of the 2,4,6-tri-tert-butylphenyl radical to 3,5-di-tert-butylneophyl, which has a short but strongly curved reaction path, was investigated using instanton theory. We found the tunneling path to deviate qualitatively from the classical intrinsic reaction coordinate, the steepest-descent path in mass-weighted Cartesian coordinates. To perform that comparison, we implemented a new variant of the predictor-corrector algorithm for the calculation of the intrinsic reaction coordinate. We used the reaction force analysis method as a means to decompose the reaction barrier into structural and electronic components. Due to the narrow energy barrier, atom tunneling is important in the abovementioned reaction, even above room temperature. Our calculated rate constants between 350 K and 100 K agree well with experimental values. We found a H/D kinetic isotope effect of almost 106 at 100 K. Tunneling dominates the protium transfer below 400 K and the deuterium transfer below 300 K. We compared the lengths of the tunneling path and the classical path for the hydrogen atom transfer in the reaction HCl + Cl and quantified the corner cutting in this reaction. At low temperature, the tunneling path is about 40% shorter than the classical path.


 Please wait while we load your content...
Please wait while we load your content...