Analyzing the dependence of oxygen incorporation current density on overpotential and oxygen partial pressure in mixed conducting oxide electrodes
Abstract
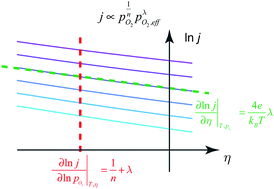
The oxygen incorporation reaction, which involves the transformation of an oxygen gas molecule to two lattice oxygen ions in a mixed ionic and electronic conducting solid, is a ubiquitous and fundamental reaction in solid-state electrochemistry. To understand the reaction pathway and to identify the rate-determining step, near-equilibrium measurements have been employed to quantify the exchange coefficients as a function of oxygen partial pressure and temperature. However, because the exchange coefficient contains contributions from both forward and reverse reaction rate constants and depends on both oxygen partial pressure and oxygen fugacity in the solid, unique and definitive mechanistic assessment has been challenging. In this work, we derive a current density equation as a function of both oxygen partial pressure and overpotential, and consider both near and far from equilibrium limits. Rather than considering specific reaction pathways, we generalize the multi-step oxygen incorporation reaction into the rate-determining step, preceding and following quasi-equilibrium steps, and consider the number of oxygen ions and electrons involved in each. By evaluating the dependence of current density on oxygen partial pressure and overpotential separately, one obtains the reaction orders for oxygen gas molecules and for solid-state species in the electrode. We simulated the oxygen incorporation current density-overpotential curves for praseodymium-doped ceria for various candidate rate-determining steps. This work highlights a promising method for studying the exchange kinetics far away from equilibrium.


 Please wait while we load your content...
Please wait while we load your content...