Unifying the microscopic picture of His-containing turns: from gas phase model peptides to crystallized proteins†
Abstract
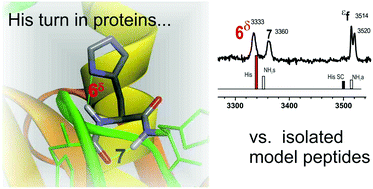
The presence in crystallized proteins of a local anchoring between the side chain of a His residue, located in the central position of a γ- or β-turn, and its local main chain environment, was assessed by the comparison of protein structures with relevant isolated model peptides. Gas phase laser spectroscopy, combined with relevant quantum chemistry methods, was used to characterize the γ- and β-turn structures in these model peptides. A conformer-selective NH stretch infrared study provided evidence for the formation in vacuo of two types of short-range H-bonded motifs, labelled ε-6δ and δ-δ7/πH, bridging the His side chain (in its gauche+ rotamer) to the neighbouring NH(i) and CO(i) sites of the backbone; each side chain-backbone motif was found to be specific of the tautomer (ε or δ) adopted by the His side chain in its neutral form. A close comparison between β- and γ-turns, selected from the Protein Data Bank, and the gas phase models demonstrated that a significant proportion of the gauche+ His rotamer distribution of proteins was well described by the corresponding gas phase H-bonded structures. This is consistent with the persistence of local 6δ and δ7/πH intramolecular interactions in proteins, emphasizing the relevance of gas phase data to secondary structures that are poorly accessible to solvents, e.g., in the case of a specific compact topology (Xxx-His β-turns). Deviations from the gas phase structures were also observed, mainly in His-Xxx β-turns, and assigned to solvent accessible turn structures. They were well accounted for by theoretical models of microhydrated turns, in which a few solvent molecules take over the gas phase motifs, constituting a water-mediated local anchoring of the His side chain to the backbone. Finally, the present gas phase benchmark models also pinpointed weaknesses in the protein structure determination by X-ray diffraction analysis; in particular, besides the lack of tautomer information, inaccuracies in the description of imidazole ring flip rotamerism were identified.


 Please wait while we load your content...
Please wait while we load your content...