Tunnelling and barrier-less motions in the 2-fluoroethanol–water complex: a rotational spectroscopic and ab initio study†
Abstract
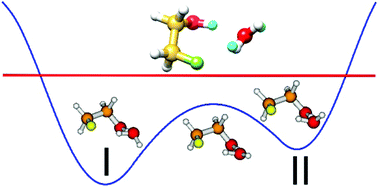
The pure rotational spectrum of the 2-fluoroethanol (2-FE)⋯water complex was measured using a chirped pulse Fourier-transform microwave spectrometer and a cavity-based Fourier-transform microwave spectrometer. In the detected 2-FE⋯water conformer, 2-FE serves as a proton donor, in contrast to its role in the observed ethanol⋯water conformer, while water acts simultaneously as a hydrogen bond donor and acceptor, forming a hydrogen-bonded ring with an OH⋯O and an OH⋯F hydrogen bond. Comparison to the calculated dipole moment components suggests that the observed structure sits between the two most stable minima identified theoretically. This conclusion is supported by extensive deuterium isotopic data. Further analysis shows that these two minima are connected by a barrier-less wagging motion of the non-bonded hydrogen of the water subunit. The observed narrow splitting with a characteristic 3 : 1 intensity ratio is attributed to an exchange of the bonded and non-bonded hydrogen atoms of water. The tunneling barrier of a proposed tunneling path is calculated to be as low as 5.10 kJ mol−1. A non-covalent interaction analysis indicates that the water rotation motion along the tunneling path has a surprisingly small effect on the interaction energy between water and 2-FE.
- This article is part of the themed collection: CSC100: Celebrating Canadian Chemistry


 Please wait while we load your content...
Please wait while we load your content...