Acceptor doping in the proton conductor SrZrO3
Abstract
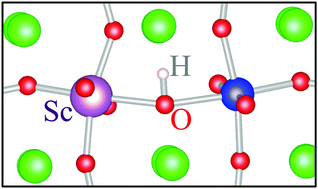
Perovskite zirconates such as SrZrO3 exhibit improved proton solubility and conductivity when doped with trivalent cations substituting at the Zr site. In this work, we present a detailed study of Sc and Y dopants in SrZrO3 based on first-principles, hybrid density-functional calculations. When substituting at the Zr site (ScZr, YZr), both dopants give rise to a single, deep acceptor level, where the neutral impurity forms a localized hole polaron state. The ε(0/−) charge transition levels are 0.60 eV and 0.58 eV above the valence-band maximum for ScZr and YZr, respectively. Under certain growth conditions, Sc and Y will form self-compensating donor species by substituting at the Sr site (ScSr, YSr), and this is detrimental to proton conductivity. Due to its larger ionic radius, Y exhibits a greater tendency than Sc to self-compensate at the Sr site. We also investigated the proton–dopant association. The binding energy of a proton to a negatively charged acceptor impurity is 0.41 eV for Sc and 0.31 eV for Y, indicating that proton transport is limited by trapping at impurity sites.


 Please wait while we load your content...
Please wait while we load your content...