Modulating the electronic structure of lanthanum manganite by ruthenium doping for enhanced photocatalytic water oxidation†
Abstract
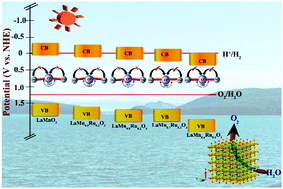
To the best of our knowledge this is the first report in which ruthenium doped polycrystalline lanthanum manganite, LaMn1−xRuxO3 (x = 0.0–0.4), having high efficacy for oxygen production from water without the use of any sacrificial reagent or co-catalyst and as an efficient photocatalyst for dye degradation is reported. Ruthenium doping alters the crystal structure of the parent LaMnO3 (LMO) due to the induced chemical pressure of the larger Ru4+ ion, which facilitates a bond angle of 180° in the Mn3+–O–Mn4+ plane resulting in the easy extraction of a photo-generated charge carrier population leading to enhanced photocatalytic activity. Rietveld refinements reveal that the parent compound LMO crystallizes in the rhombohedral phase, while upon an increase in the doping concentration of ruthenium, the phase of the compounds changes from the rhombohedral to the cubic phase. The percentage contribution of each phase has been estimated using the sixth-order polynomial and pseudo-Voigt function. Typically, all the compositions, LaMn1−xRuxO3 (x = 0.0–0.4), were prepared by a conventional solid state route and studied for their photocatalytic activity. The synthesized compounds were investigated by powder X-ray diffraction (PXRD), UV-visible diffuse reflectance spectroscopy (DRS), Fourier transform infrared spectroscopy (FTIR), field emission scanning electron microscopy (FESEM), energy-dispersive X-ray spectroscopy (EDX) and X-ray photoelectron spectroscopy (XPS) analysis. The structure–property correlation of the compound is presented based on Rietveld refinement combined with the experimental data. The as-prepared compounds show efficient photocatalytic oxygen gas production from water without the use of any co-catalyst or sacrificial reagents. Among the five compositions, LaMn0.7Ru0.3O3 shows the highest O2 production efficiency (4.73 mmol g−1 h−1) with an apparent quantum yield (AQY) of 7.43%. These ruthenium doped compositions also exhibit superior dye degradation properties, studied by taking the industrial dye methyl orange (MO) as the model compound.


 Please wait while we load your content...
Please wait while we load your content...