Water-repellent hydrophilic nanogrooves
Abstract
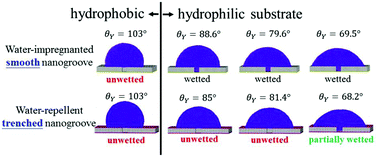
The wetting behavior of a nanodrop atop a nanogroove on a smooth or a rough surface is explored by many-body dissipative particle dynamics and Surface Evolver. The nanogroove possesses the same contact angle (θY) as that of the surface. Depending on whether the groove is initially wetted or not, two critical contact angles 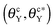 beyond which the groove cannot be wetted are determined. Three regimes are identified: (i) as θY ≤ θcY, the groove is always wetted; (ii) as
beyond which the groove cannot be wetted are determined. Three regimes are identified: (i) as θY ≤ θcY, the groove is always wetted; (ii) as 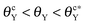 , both impregnated and unwetted states can be observed; (iii) as
, both impregnated and unwetted states can be observed; (iii) as 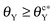 , the groove cannot be impregnated. As the drop volume is increased, both θcY and
, the groove cannot be impregnated. As the drop volume is increased, both θcY and 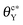 decrease but become insensitive to the volume eventually. Surface roughness tends to hamper the impregnation of grooves by liquid. Compared to a smooth surface, both critical contact angles of a rough surface with regular shallow pits are smaller. As a result, a large drop is unable to wet the groove with a rough surface even when the surface becomes slightly hydrophilic. When the surface structure within the groove is modified from shallow pits to straight trenches, the critical contact angle is further reduced. Our simulation outcomes show that the surface structure within the groove is crucial for liquid imbibition and it is possible to fabricate hydrophilic cavities that can prevent impregnation, without resorting to chemical modification processes.
decrease but become insensitive to the volume eventually. Surface roughness tends to hamper the impregnation of grooves by liquid. Compared to a smooth surface, both critical contact angles of a rough surface with regular shallow pits are smaller. As a result, a large drop is unable to wet the groove with a rough surface even when the surface becomes slightly hydrophilic. When the surface structure within the groove is modified from shallow pits to straight trenches, the critical contact angle is further reduced. Our simulation outcomes show that the surface structure within the groove is crucial for liquid imbibition and it is possible to fabricate hydrophilic cavities that can prevent impregnation, without resorting to chemical modification processes.


 Please wait while we load your content...
Please wait while we load your content...