Identification of the smallest peptide with a zwitterion as the global minimum: a first-principles study on arginine-containing peptides†
Abstract
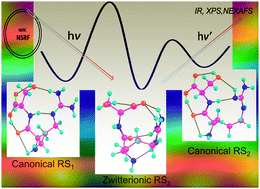
Zwitterions are believed to play an important role in determining the structures, properties and functions of peptides and proteins. However, the smallest peptide with a zwitterionic structure as the global minimum in the gas phase is still not yet identified. In this study, an effective step-by-step strategy has been used to characterize the stable conformers of arginine-containing peptides arginylalanine (ArgAla) and arginylserine (ArgSer). Energy calculations at the DSD-PBEP86-D3BJ/aug-cc-pVTZ level and further extrapolation to the complete basis set (CBS) limit have confirmed, for the first time, that ArgSer appears to be a promising candidate as the smallest peptide with a zwitterionic global minimum structure. First-principles simulations have been performed for near-edge X-ray absorption fine-structure (NEXAFS) spectra and X-ray photoelectron spectra (XPS) at C, N and O K-edges, as well as for infrared (IR) spectra of these arginine-containing peptides. Notable spectral differences were found which enable the unambiguous identification of different neutral forms in future experiments. Our study thus provides valuable insights into the structural stability of zwitterions with the increase of molecular size and illustrates the competition between the canonical and zwitterionic isomers.


 Please wait while we load your content...
Please wait while we load your content...