Photoelectrochemical water reduction over wide gap (Ag,Cu)(In,Ga)S2 thin film photocathodes†
Abstract
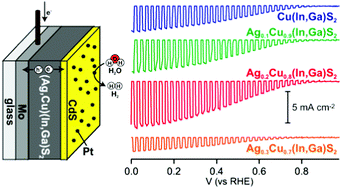
The effects of partial replacement of Cu with Ag in a Cu(In,Ga)S2 (CIGS) thin film on its structural, optical, electrostructural, and photoelectrochemical (PEC) properties were investigated, in order to improve its performance for PEC water reduction under sunlight illumination. Results from X-ray diffraction (XRD) analyses revealed the successful partial replacement of Cu with Ag to form solid-solutions with different Ag/(Ag + Cu) ratios (A(x)CIGS, x = Ag/(Ag + Cu) = 0.1, 0.2, 0.3 and 0.4), as confirmed by a gradual change in the (112) reflections to smaller 2θ angles with increasing Ag/(Ag + Cu) ratio. Analyses of the photoabsorption properties of the materials using photoacoustic spectroscopy indicated changes in the band gap energies associated with increasing the Ag/(Ag + Cu) ratio. In addition, valence band maximum potentials of A(x)CIGS were deepened gradually with increasing Ag/(Ag + Cu) ratio. After modifying these A(x)CIGS films with a CdS ultrathin (ca. 70 nm) layer and a Pt catalyst, the PEC water reduction properties were evaluated in an electrolyte solution with the pH adjusted to 6.5, under simulated sunlight (AM 1.5G) radiation. Compared to the CdS- and Pt-modified Ag-free A(x)CIGS (A(0)CIGS) films, appreciable enhancements in the PEC properties were observed for electrodes based on A(x)CIGS (x > 0) films, and the best PEC performance was obtained for the electrode based on the A(0.2)CIGS film. However, the electrode derived from the A(x)CIGS film with Ag/(Ag + Cu) ratios higher than 0.3 showed diminished PEC properties due to the partial conversion of its semiconducting properties from p-type to n-type.


 Please wait while we load your content...
Please wait while we load your content...