Interactions of ionic liquids and surfaces of graphene related nanoparticles under high pressures†
Abstract
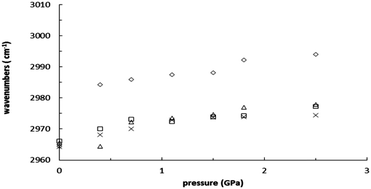
High-pressure infrared spectroscopy was used to study the interactions between 1-methyl-3-propylimidazolium iodide [MPIM]I and graphene-based nanoparticles. The results obtained at ambient pressure indicate the imidazolium ring of the cation to be a more favorable moiety for adsorption than alkyl C–H groups at ambient pressure. Upon increasing the pressure, the dominant C2–H band of pure [MPIM]I yields a significant red frequency shift. As the mixtures, i.e., graphene oxide (GO)/[MPIM]I, reduced graphene oxide (RGO)/[MPIM]I, and graphene (G)/[MPIM]I, were compressed, mild shifts in the C2–H absorption frequency were observed. The absence of drastic red-shifts suggests that the local C2–H structures may be perturbed by the addition of GO, RGO, and G under high pressures. When pure [MPIM]I was compressed from ambient to 0.4 GPa, the alkyl C–H band at ca. 2964 cm−1 was blue-shifted to 2984 cm−1. This discontinuous jump occurring around 0.4 GPa becomes less obvious for the mixtures GO/[MPIM]I, RGO/[MPIM]I, and G/[MPIM]I. The results of this study suggest that the addition of GO, RGO, and G can disturb the local structures of alkyl C–H under high pressures, demonstrating that high pressures may have the potential to tune the strength of ionic liquid–surface interactions and the performance of energy storage devices (e.g. supercapacitors).


 Please wait while we load your content...
Please wait while we load your content...