Direct experimental probing and theoretical analysis of the reaction between the simplest Criegee intermediate CH2OO and isoprene†
Abstract
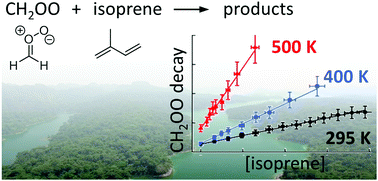
Recent advances in the spectroscopy of Criegee intermediates (CI) have enabled direct kinetic studies of these highly reactive chemical species. The impact of CI chemistry is currently being incorporated into atmospheric models, including their reactions with trace organic and inorganic compounds. Isoprene, C5H8, is a doubly-unsaturated hydrocarbon that accounts for the largest share of all biogenic emissions around the globe and is also a building block of larger volatile organic compounds. We report direct measurements of the reaction of the simplest CI (CH2OO) with isoprene, using time-resolved cavity-enhanced UV absorption spectroscopy. We find the reaction to be pressure-independent between 15–100 Torr, with a rate coefficient that varies from (1.5 ± 0.1) × 10−15 cm3 molecule−1 s−1 at room temperature to (23 ± 2) × 10−15 cm3 molecule−1 s−1 at 540 K. Quantum chemical and transition-state theory calculations of 16 unique channels for CH2OO + isoprene somewhat underpredict the observed T-dependence of the total reaction rate coefficient, but are overall in good agreement with the experimental measurements. This reaction is broadly similar to those with smaller alkenes, proceeding by 1,3-dipolar cycloaddition to one of the two conjugated double bonds of isoprene.


 Please wait while we load your content...
Please wait while we load your content...