Unusual molecular mechanism behind the thermal response of polypeptoids in aqueous solutions†
Abstract
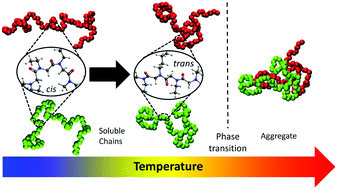
Poly(α-peptoid)s, a structural isomer to polypeptides, have recently attracted a significant amount of scientific attention. However, the molecular mechanism behind the thermal response of this class of polymers is unknown. Here, the thermal response of two polypeptoids in aqueous solutions was studied by different methodologies, including dynamic light scattering, IR spectroscopies, NMR, etc. Our studies focused on two polypeptoids with identical alkyl side chain compositions, but different architecture; i.e., cyclic and linear. Aqueous solutions of the cyclic and linear polymers present phase transition temperatures at 43 °C and 47 °C, respectively, that have an anomalous dependence on the polymer morphology as expected from macromolecules having very similar solvent interactions, but different conformational entropy. The atypical trend in the phase transition temperature is found to be caused by the initiator required in the synthesis which favors the formation of soluble dimers in the cyclic polymer. Our experimental findings also demonstrate that the phase transition, irrespective of the morphology, is governed by the polymer backbone conformation which depends on the composition and structure of the alkyl side chains. This proposed mechanism is novel and different from the commonly assumed mechanism for thermo-responsive polymers in which the hydration of the polymer cause by a coil to globule transition is the determining factor. Moreover, the proposed mechanism is likely to be general since it can explain not only the experimental findings of this work, but also observations of the thermal response and conformation of other studied polypeptoids in water. Finally, our mechanism gives a molecular framework for the rational designed of polypeptoids with tailored phase transition temperatures.


 Please wait while we load your content...
Please wait while we load your content...