A conformational study of protonated noradrenaline by UV–UV and IR dip double resonance laser spectroscopy combined with an electrospray and a cold ion trap method†
Abstract
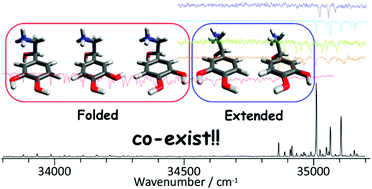
The conformer-selected ultraviolet (UV) and infrared (IR) spectra of protonated noradrenaline were measured using an electrospray/cryogenic ion trap technique combined with photo-dissociation spectroscopy. By comparing the UV photo dissociation (UVPD) spectra with the UV–UV hole burning (HB) spectra, it was found that five conformers coexist under ultra-cold conditions. Based on the spectral features of the IR dip spectra of each conformer, two different conformations on the amine side chain were identified. Three conformers (group I) were assigned to folded and others (group II) to extended structures by comparing the observed IR spectra with the calculated ones. Observation of the significantly less-stable extended conformers strongly suggests that the extended structures are dominant in solution and are detected in the gas phase by kinetic trapping. The conformers in each group are assignable to rotamers of OH orientations in the catechol ring. By comparing the UV–UV HB spectra and the calculated Franck–Condon spectra obtained by harmonic vibrational analysis of the S1 state, with the aid of relative stabilization energies of each conformer in the S0 state, the absolute orientations of catechol OHs of the observed five conformers were successfully determined. It was found that the 0–0 transition of one folded conformer is red-shifted by about 1000 cm−1 from the others. The significant red-shift was explained by a large contribution of the πσ* state to S1 in the conformer in which an oxygen atom of the meta-OH group is close to the ammonium group.
- This article is part of the themed collection: Bunsentagung 2017: Physical Chemistry for Life Sciences


 Please wait while we load your content...
Please wait while we load your content...