Ultrafast energy and electron transfers in structurally well addressable BODIPY-porphyrin-fullerene polyads†
Abstract
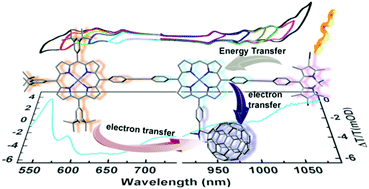
Two electron transfer polyads built upon [C60]–[ZnP]–[BODIPY] (1) and [ZnP]–[ZnP](–[BODIPY])(–[C60]) (2), where [C60] = N-methyl-2-phenyl-3,4-fulleropyrrolidine, [BODIPY] = boron dipyrromethane, and [ZnP] = zinc(II) porphyrin, were synthesized along with their corresponding energy transfer polyads [ZnP]–[BODIPY] (1a) and [ZnP]–[ZnP]–[BODIPY] (2a) as well as relevant models. These polyads were studied using cyclic voltammetry, DFT computations, steady state and time-resolved fluorescence spectroscopy, and fs transient absorption spectroscopy. The rates for energy transfer, kET, [BODIPY]* → [ZnP] are ∼2.8 × 1010 s−1 for both 1a and 2a, with an efficiency of 99%. Concurrently, the fast appearance of the [C60]−˙ anion for 1 and 2 indicates that the charge separation occurs on the 20–30 ps timescale with the rates of electron transfer, ket, [ZnP]*/[C60] → [ZnP]+˙/[C60]−˙ of ∼0.9 × 1010 to ∼3.8 × 1010 s−1. The latter value is among the fastest for these types of polyads. Conversely, the charge recombination operates on the ns timescale. These rates are comparable to or faster than those reported for other more flexible [C60]–[ZnP]–[BODIPY] polyads, which can be rationalized by the donor–acceptor separations.


 Please wait while we load your content...
Please wait while we load your content...