Polymorphism of the quasiracemate d-2-aminobutyric acid:l-norvaline†
Abstract
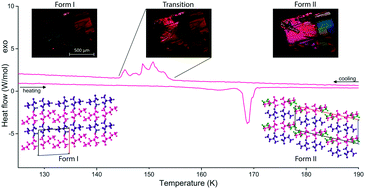
Here we report two new crystal structures of the amino acid quasiracemate D-2-aminobutyric acid:L-norvaline, as well as the fully reversible solid-state phase transition which connects the two forms. The room temperature form II is obtained by co-crystallisation through slow evaporation from an equimolar aqueous solution. Form I is obtained through cooling of form II below 150 K. The crystal structures of forms I and II have been determined using single crystal X-ray diffraction at 100 and 150 K, respectively. Both forms consist of 2D hydrogen bonded bilayers with an LD–LD hydrogen bonding pattern. The room temperature form II shows two conformations for the L-norvaline side chain in a disordered 50/50 occupancy distribution. The solid-state phase transition between the two polymorphic forms involves a displacement of the bilayers and conformational changes in all molecules. It has been further characterised using differential scanning calorimetry and thermal polarisation microscopy. These crystal structures and the single-crystal-to-single-crystal phase transition are similar to other quasiracemates and racemates of linear chain aliphatic amino acids.


 Please wait while we load your content...
Please wait while we load your content...