Facile and economical synthesis of ZnS nanotubes and their superior adsorption performance for organic dyes†
Abstract
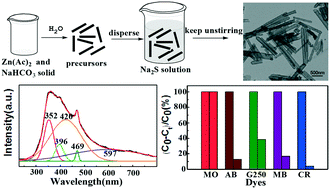
Developing a green and economical synthetic strategy for ZnS nanotubes is of great interest. In this work, ZnS nanotubes are successfully fabricated using only three raw materials (Zn(CH3COO)2, NaHCO3 and Na2S) through a simple two step synthetic route at ambient temperature. It is worth noting that the process is economical and environmentally friendly, without the need for high pressure or elevated temperature conditions. Interestingly, a very obvious feature is the existence of abundant sulfur surface defects on the as-prepared ZnS nanotubes, which not only produces strong defect-related fluorescence properties, but also leads to abundant water adsorbed on the surface of zinc sulfide. Furthermore, the as-prepared ZnS nanotubes as an adsorbent are exploited in the adsorption of organic dyes. Notably, the resultant products display superior selective adsorption ability for anionic dyes with amine (–NH2/NH) functional groups over a wide pH range through the synergistic effect of hydrogen bonding and electrostatic attraction between the dye molecules and the adsorbent. Taking CR dye as an example, the maximum adsorption capacity for the removal of Congo red reaches 724.6 mg g−1 in water, much higher than many composite adsorbents reported in the literature. Thus, the present study not only presents a promising strategy for fabrication of ZnS nanotubes but also indicates their great potential application as environmentally friendly adsorbents in organic dye separation technologies or dye removal.


 Please wait while we load your content...
Please wait while we load your content...