Heat-up and gram-scale synthesis of Cu-poor CZTS nanocrystals with controllable compositions and shapes†
Abstract
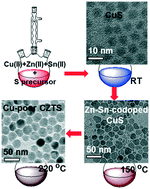
We report the gram-scale synthesis of Cu-poor CZTS nanocrystals (NCs) with tuned shapes and compositions by using a facile phosphine-free heat-up procedure. Based on the detailed characterization and analyses of the evolution of crystal phases, compositions, morphologies and optical spectra over the reaction stage and temperature, we explore the reaction mechanism. The synthesis reaction involves the formation of binary CuS (covellite) nanoparticles at room temperature, and the conversion from the as-formed CuS to Zn–Sn-codoped CuS NCs as intermediates at 150 °C and finally to quaternary CZTS NCs in the kesterite phase at 220 °C, during which the NCs gradually grow due to the progressive incorporation of foreign cations (Zn2+ and Sn2+) into the as-formed CuS NCs. The introduction of dodecanethiol (DT) leads to the formation of bullet-like CZTS NCs in the wurtzite phase. The Cu-poor CZTS synthesis is reproducible by upscaling the amounts of chemicals and solvents/ligands, which allows for a promising approach to gram-scale production of Cu-based chalcogenides for potential applications in photovoltaics.


 Please wait while we load your content...
Please wait while we load your content...