Spherically aggregated Cu2O–TA hybrid sub-microparticles with modulated size and improved chemical stability†
Abstract
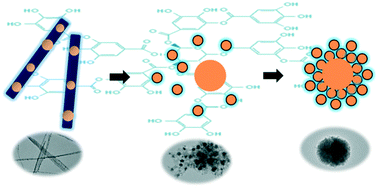
In this paper, tannic acid (TA) was used to assist the fabrication of spherically aggregated Cu2O–TA hybrid sub-microparticles with good chemical stability and controllable sizes. TA exhibited multiple functions in the synthesis: the reductant for the formation of Cu2O crystal seeds, the stabilizer of the primary Cu2O crystals and the protector against the oxidation of Cu2O. A probable mechanism was presumed, including 1) seed formation and TA encapsulation, 2) primary crystal nucleation and growth, and 3) aggregation into spheres. We found that pH played a key role in determining the final morphology of the product while the precursor concentration strongly influenced the size of the particles. At a suitable pH, hierarchically structured Cu2O–TA sub-microspheres with nanocrystals as building blocks were produced. The size of the spheres can be tuned from 130 to 670 nm by adjusting the precursor concentration. Cu2O–TA hybrids exhibited excellent chemical stability against water and oxygen. The method reported here possessed many advantages like cost-effectiveness, facility of preparation, environmental friendliness, chemical stability and excellent adjustability. The properties of the prepared Cu2O particles might facilitate their applications in marine antifouling paints, adsorption materials and antibacterial agents.


 Please wait while we load your content...
Please wait while we load your content...