Diverse binding of important anions in 1-D tricopper anion coordination polymer (ACP) architectures†‡§
Abstract
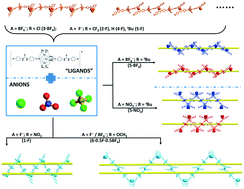
With the idea of supramolecular hosts encircling monoanions, a series of urea-containing trinuclear cationic copper complexes were designed, prepared and structurally analyzed. Copper(I) acetylides [Cu3(μ-dppm)3(μ3-η1-C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CC6H4-4-NHC(O)NHC6H4-4-R)2]+ (dppm = bis(diphenylphosphino)methane; R = NO2 (1), CF3 (2), Cl (3), H (4), tBu (5), and OCH3 (6)) were prepared with F−, BF4−, or NO3− present as the anion. Eight 1-D anion coordination polymers (ACPs), 1·F, 2·F, 3·BF4, 4·F, 5·F, 5·BF4, 5·NO3 and 6·0.5F·0.5BF4, were clearly determined by X-ray diffraction. Diversity in the polymeric architecture is generated by both the choice of R-group used and anion present. 2·F, 3·BF4, 4·F, and 5·F show linear 1-D structures; 5·BF4 and 5·NO3 feature single-stranded helical structures; 1·F and 6·0.5F·0.5BF4 exhibit meso-helical structures. Solid state UV-vis absorption and luminescence properties are also discussed.
CC6H4-4-NHC(O)NHC6H4-4-R)2]+ (dppm = bis(diphenylphosphino)methane; R = NO2 (1), CF3 (2), Cl (3), H (4), tBu (5), and OCH3 (6)) were prepared with F−, BF4−, or NO3− present as the anion. Eight 1-D anion coordination polymers (ACPs), 1·F, 2·F, 3·BF4, 4·F, 5·F, 5·BF4, 5·NO3 and 6·0.5F·0.5BF4, were clearly determined by X-ray diffraction. Diversity in the polymeric architecture is generated by both the choice of R-group used and anion present. 2·F, 3·BF4, 4·F, and 5·F show linear 1-D structures; 5·BF4 and 5·NO3 feature single-stranded helical structures; 1·F and 6·0.5F·0.5BF4 exhibit meso-helical structures. Solid state UV-vis absorption and luminescence properties are also discussed.


 Please wait while we load your content...
Please wait while we load your content...