Solid-state inclusion of C60 and C70 in a co-polymer induced by metal–ligand coordination of a Zn–porphyrin cage with a bis-pyridyl perylene derivative†
Abstract
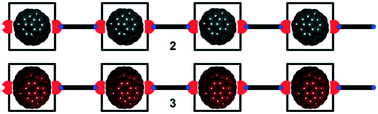
We describe three unprecedented solid-state crystal structures (1–3) involving two (1) or three (2 & 3) different components: a suitable bis-porphyrin cage, a bis-pyridyl perylene derivative that is too large to bind the cage in a ditopic manner and, for 2 and 3, a fullerene (C60 and C70, respectively). Single co-crystals of the two/three components, suitable for X-ray diffraction analyses, grew from their equimolar mixture in p-xylene solution. Remarkably, the co-polymeric 1D chains that are self-assembled in the solid state possess a metal–organic framework (MOF). The fullerene, C60 (2) or C70 (3), is included in all repeating monomeric units of the Zn(II)–bisporphyrin component, which functions as a molecular cage. The formed 1 : 1 inclusion complexes exhibit a parallel arrangement of the two porphyrin units. In turn, adjacent 1 : 1 inclusion complexes are connected through bis-pyridyl perylene ligands acting as pillars. The exo-axial coordination of Zn-centers with the nitrogen atoms of the pyridyl residues defines a new motif of perylene derivatives sandwiched between the 1 : 1 inclusion complexes. The included fullerenes C60/C70 interact with the porphyrin walls of the cages and their aliphatic spacer chains. In particular, the interactions of Zn(II)–bisporphyrin⋯C60/C70 observed in the 1 : 1 inclusion complexes were analysed using Density Functional Theory (DFT) calculations (PB86-D3/def2-TZVP level of theory) and Molecular Electrostatic Potential (MEP) surfaces, and characterized by means of Bader's theory of “atoms-in-molecules” (AIM). The computation of the energetic features of the diverse noncovalent interactions including Zn–porphyrin⋯π(fullerene) and C–H⋯π(fullerene) interactions revealed their conspicuous role in the stabilization of the one-dimensional supramolecular frameworks for both solid-state crystal structures.
- This article is part of the themed collection: Covalent organic frameworks and organic cage structures


 Please wait while we load your content...
Please wait while we load your content...