MOF catalysts in biomass upgrading towards value-added fine chemicals†
Abstract
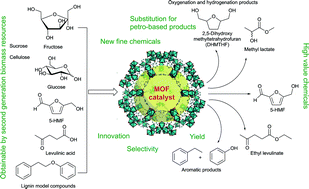
The development of new synthetic routes from biomass sources towards already existing molecules, which are then called bio-based molecules, or the transformation of biomass into new building blocks and materials will be of great impact. The review presents a critical comparison between metal–organic frameworks (MOFs) and other catalysts (e.g. zeolites) for biomass transformation and valorization to platform chemicals: cellulose hydrolysis to glucose, fructose or sorbitol; fructose, glucose or maltose to 5-hydroxymethylfurfural (5-HMF); sucrose to methyl lactate; furans, levulinic acid, lignin or vanillin as feedstock; triglycerides to esters and glycerol. For example, in the case of cellulose hydrolysis as well as glucose isomerization MOF-based catalysts could not compete with zeolites and sulfonated carbon which display significantly higher activity. In DMSO, MIL-101Cr-SO3H-15% and NUS-6(Hf) are among the best heterogeneous catalysts reported so far for the conversion of fructose into 5-HMF. For the glucose-to-5-HMF transformation MIL-101Cr-SO3H is only a low-to-medium activity catalyst for 5-HMF while mesoporous tantalum phosphate as well as Sn montmorillonite display significantly higher activities. On the other hand, MIL-101Cr-SO3H preferentially transformed glucose to 5-HMF over levulinic acid while the catalysts Amberlyst-15 and sulfuric acid gave mostly levulinic acid. For levulinic acid conversion to ethyl levulinate UiO-66Zr catalysts can compete with other heterogeneous catalysts for the levulinic esterification reaction. For active MOF catalysts open metal sites (coordinatively unsaturated sites) are important as the activity increases with the amount of missing linkers. The two MOFs MIL-101Cr and UiO-66 and their derivatives are used in many studies. These MOFs did not only act as catalysts themselves but also served as hosts or support to embedded catalytic species, e.g., phosphotungstic acid (PTA), ruthenium and palladium nanoparticle (Ru-NP, Pd-NP) or poly(N-bromomaleimide) catalysts. For the conversion of vanillin into 2-methoxy-4-methylphenol the selectivity of Pd@UiO-66Zr-NH2 was quantitative compared with other supported Pd catalysts (selectivity 48%). Further, MOFs were used as precursors for decomposition and carbonization due to their high porosity and uniformly distributed metal centers to yield catalytically active metal–carbonaceous materials with high thermal and chemical stability. For example, metal nanoparticles supported on nanoporous carbon (M/NC) were synthesized by carbonization and carbothermal reduction of Ru, W, V, and Ti metal precursors loaded in IRMOF-1 or IRMOF-3. Fe–Co-based MOF-derived catalysts are a highly efficient system for the conversion of 5-HMF to 2,5-diformylfuran. In water-containing reactions, the water stability of MOFs is of high importance.
- This article is part of the themed collections: 2017 Highlight article collection and Metal-Organic Framework Catalysis


 Please wait while we load your content...
Please wait while we load your content...