Syntheses of potent teixobactin analogues against methicillin-resistant Staphylococcus aureus (MRSA) through the replacement of l-allo-enduracididine with its isosteres†
Abstract
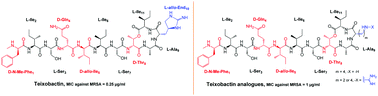
The recently discovered cyclic depsipeptide, teixobactin, is a highly potent antibiotic against multi-drug resistant pathogens such as methicillin-resistant Staphylococcus aureus (MRSA) and Mycobaterium tuberculosis. It comprises of 4 D amino acids and a rare L-allo-enduracididine amino acid. The synthesis of a properly protected L-allo-enduracididine amino acid and its incorporation into teixobactin is time consuming, synthetically challenging and low yielding and is therefore a major bottleneck in the development of potent analogues of teixobactin. In this article, we have synthesised 8 analogues of teixobactin using commercially available building blocks by replacing the L-allo-enduracididine amino acid with its isosteres. Furthermore, we have tested all the compounds against a panel of Gram positive bacteria including MRSA and explained the observed trend in biological activity. Although all the analogues were active, three analogues from this work, showed very promising activity against MRSA (MIC 1 μg mL−1). We can conclude that amino acids which are the closest isosteres of L-allo-enduracididine are the key to synthesising simplified potent analogues of teixobactin using rapid syntheses and improved yields.
- This article is part of the themed collection: Suprabiomolecular Chemistry


 Please wait while we load your content...
Please wait while we load your content...