Element strategy of oxygen evolution electrocatalysis based on in situ spectroelectrochemistry
Abstract
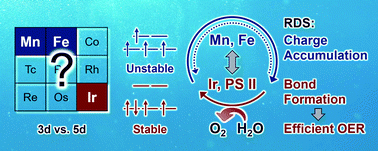
Oxygen evolution electrocatalysis has received extensive attention due to its significance in biology, chemistry, and technology. However, it is still unclear how the abundant 3d-elements can be used to drive the four-electron oxidation of water as efficiently as in Nature. In this Feature Article, we will propose a design strategy concerning the optimization of the charge accumulation process based on our ongoing spectroelectrochemical study on Mn, Fe, and Ir oxygen evolution catalysts. Spectroscopic identification of the reaction intermediates showed that the activity of MnO2 and Fe2O3 was dictated by the generation of Mn3+ and Fe4+, whereas in the case of IrOx, the activity did not correlate with the valence change of Ir. The efficiency of charge accumulation through valence change is closely linked with the spin configuration of the metal center, because charge disproportionation, which was found to inhibit charge accumulation in the high-spin 3d metals, requires an electron in the eg orbital. In addition to directly increasing the overpotential through the generation of an unstable intermediate, charge disproportionation inhibits charge accumulation by dissipating the total oxidative energy of the system. A favorable charge accumulation process may also be beneficial for electrode kinetics due to the enhanced coupling between reaction rates and electrochemical driving force. The model proposed in this study may help explain why low-spin 4d/5d rare metals are often more active than the abundant high-spin 3d materials for multi-electron transfer reactions in general, and provides new insight into how active 3d-metal catalysts can be synthesized by optimizing the energetics of both bond formation and charge accumulation.
- This article is part of the themed collection: 2017 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...