Interface modulation of bacteriogenic Ag/AgCl nanoparticles by boosting the catalytic activity for reduction reactions using Co2+ ions†
Abstract
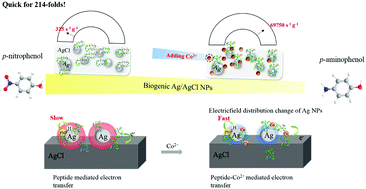
Boosting the low catalytic activity of bacteriogenic Ag/AgCl nanoparticles (NPs) by adding Co2+ in a model reaction, i.e. p-nitrophenol (PNP) reduction, was observed. Under optimal conditions, Co–Ag/AgCl NPs show an apparent rate constant (kapp) of 0.0837 s−1 upon PNP reduction. Considering the trace amount of Co–Ag/AgCl NPs used in catalysis (about 1.2 μg), the activity parameter κ (which is the ratio of kapp and catalyst mass) can reach more than 6.97 × 104 s−1 g−1, which is 214-fold higher than that of pristine Ag/AgCl NPs. Meanwhile, this κ value is also the highest value reported to date. In addition, the biogenic Co–Ag/AgCl NPs exhibit superior catalytic activities over other substrates, such as m-nitrophenol, o-nitrophenol, methyl orange and rhodamine B. The activity enhancement mechanism is supposed to be that Co2+ acts as a Lewis acid, and coordinates with the surface peptides to affect the electric field distribution at the Ag/AgCl NP interface.


 Please wait while we load your content...
Please wait while we load your content...