Analysis of changes in the glycan composition of serum, cytosol and membrane glycoprotein biomarkers of colorectal cancer using a lectin-based protein microarray
Abstract
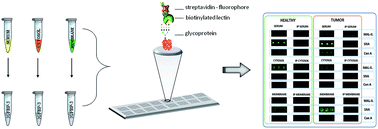
The altered glycosylation of proteins is a cancer-related marker and the monitoring of glycosylation status can significantly increase the informative value of protein biomarkers. Herein is presented an analytical method for the investigation of the glycan composition of colorectal cancer (CRC) protein biomarkers applied to different sample types using a lectin-based microarray platform. The samples included sera (from healthy persons and patients with CRC), cytosol/membrane protein fractions (from non-tumor and tumor colon tissue), and insulin-like growth factor-binding protein 3 (IGFBP-3) isolated from sera and cytosol/membrane fractions. All samples were spotted into arrays on a microarray slide, and were then incubated with a panel of 14 biotinylated lectins and a fluorescent conjugate of streptavidin. The signal intensities were detected using a microarray scanner. Statistically significant differences (Mann–Whitney test, P < 0.05) in signal intensities were found between the two groups of serum samples (with stronger signal intensities from Sambucus nigra lectin for the samples from patients with CRC than in samples from healthy individuals) and between the two groups of samples containing IGFBP-3 isolated from serum (with stronger signals from Maackia amurensis lectin-II and weaker signals from Aleuria aurantia lectin for the samples from patients with CRC). Weaker signal intensities from Aleuria aurantia lectin were observed also for samples of IGFBP-3 isolated from tumor cell membranes than for IGFBP-3 samples isolated from non-tumor tissue. The described method is applicable to the fast and high-throughput glyco-recognition analysis of differences in glycosylation patterns in various types of samples containing glycoprotein biomarkers.


 Please wait while we load your content...
Please wait while we load your content...