A novel non-enzymatic glucose sensor based on a Cu-nanoparticle-modified graphene edge nanoelectrode†
Abstract
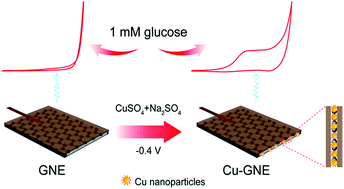
A novel, stable and sensitive non-enzymatic glucose sensor was developed by potentiostatically electrodepositing metallic Cu nanoparticles on a linear graphene edge nanoelectrode (Cu–GNE). The electrochemical performance of the Cu–GNE for the detection of glucose was investigated by cyclic voltammetry and chronoamperometry. The Cu–GNE displayed a synergistic effect of copper nanoparticles and the edge plane of the graphene sheet towards the oxidation of glucose in alkaline solution, showing a large oxidation current and negative shift in peak potential. At a detection potential of 0.5 V, the Cu–GNE sensor exhibited a wide linear range up to 1 mM glucose, with a detection limit of 0.12 μM (S/N = 3). In addition, the sensor responded very quickly (<2 s) upon the addition of glucose. Furthermore, the Cu–GNE exhibited high stability and selectivity to glucose. Poisoning by chloride ions and interference from the oxidation of common interfering species (ascorbic, dopamine, uric acid and carbohydrate) were effectively avoided. The Cu–GNE allowed highly selective and sensitive, stable and fast amperometric sensing of glucose, which is promising for the development of a non-enzymatic glucose sensor.


 Please wait while we load your content...
Please wait while we load your content...