Dynamic solid-phase RNA extraction from a biological sample in a polyester-toner based microchip
Abstract
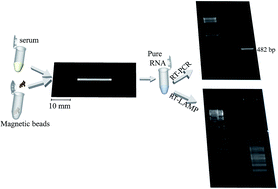
The standard molecular diagnostic techniques for dengue virus infection involve sample preparation (RNA extraction) followed by reverse transcription-nucleic acid amplification and separation/detection of DNA fragments. Efficient and reliable RNA extraction is crucial for all downstream steps. Solid-phase extraction (SPE) is one of the most used methodologies for RNA purification, and it has been largely adapted for microfluidic applications. Here, we describe a methodology for RNA purification of dengue virus in polyester-toner (PeT) microfluidic devices using magnetic silica-based dynamic solid phase extraction (dSPE). PeT microdevices were produced by a print, cut, and laminate (PCL) methodology previously described by Duarte et al. (G. R. M. Duarte, C. W. Price, B. H. Augustine, E. Carrilho and J. P. Landers, Anal. Chem., 2011, 83, 5182–5189.) The dSPE methodology involved performing adsorption, washing, and elution steps. The PeT microchip has already proven to be biocompatible and capable of performing various steps of genetic analysis involving DNA. In this work, we show for the first time that PeT microdevices can also be used for RNA analysis. High-quality pure RNA (amplifiable via RT-PCR and/or RT-LAMP) was obtained in dSPE experiments carried out in PeT microchips, demonstrating that RNA is compatible with reagents used in dSPE and with substrates used in PeT microchip fabrication. Dengue virus RNA was purified from human serum samples using the proposed dSPE methodology in PeT microchips, making this a viable technique for isolating RNA for future integration with downstream microfluidic analysis, such as RT-PCR or RT-LAMP, to produce a fully integrated microfluidic device for genetic analysis involving RNA.
- This article is part of the themed collection: Analytical Chemistry in South America


 Please wait while we load your content...
Please wait while we load your content...