Desktop NMR for structure elucidation and identification of strychnine adulteration†
Abstract
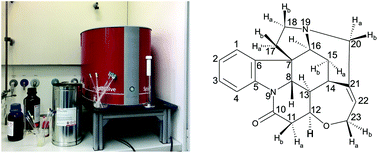
Elucidating the structure of complex molecules is difficult at low magnetic fields due to the overlap of different peak multiplets and second-order coupling effects. This is even more challenging for rigid molecules with small chemical shift differences and with prochiral centers. Since low-field NMR spectroscopy is sometimes presumed as restricted to the analysis of only small and simple molecules, this paper aims at countering this misconception: it demonstrates the use of low-field NMR spectroscopy in chemical forensics for identifying strychnine and its counterions by exploring the chemical shift as a signature in different 1D 1H and 13C experiments. Hereby the applied methodologies combine various 1D and 2D experiments such as 1D 1H, 13C, DEPT, and 2D COSY, HETCOR, HSQC, HMBC and J-resolved spectroscopy to elucidate the molecular structure and skeleton of strychnine at 1 Tesla. Strychnine is exemplified here, because it is a basic precursor in the chemistry of natural products and is employed as a chemical weapon and as a doping agent in sports including the Olympics. In our study, the molecular structure of the compound could be identified either with a 1D experiment at high magnetic field or with HMBC and HSQC experiments at 1 T. In conclusion, low-field NMR spectroscopy enables the chemical elucidation of the strychnine structure through a simple click with a computer mouse. In situations where a high-field NMR spectrometer is unavailable, compact NMR spectrometers can nevertheless generate knowledge of the structure, important for identifying the different chemical reaction mechanisms associated with the molecule. Desktop NMR is a cost-effective viable option in chemical forensics. It can prove adulteration and identify the origin of different strychnine salts, in particular, the strychnine free base, strychnine hemisulphate and strychnine hydrochloride. The chemical shift signatures report the chemical structure of the molecules due to the impact of the counterions on the chemical shift of the protons adjacent to the heteroatoms. This can serve as a methodology for the structure elucidation of complex molecules at lower-magnetic fields.


 Please wait while we load your content...
Please wait while we load your content...