Rapid infrared mapping for highly accurate automated histology in Barrett's oesophagus
Abstract
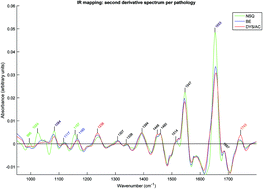
Barrett's oesophagus (BE) is a premalignant condition that can progress to oesophageal adenocarcinoma. Endoscopic surveillance aims to identify potential progression at an early, treatable stage, but generates large numbers of tissue biopsies. Fourier transform infrared (FTIR) mapping was used to develop an automated histology tool for detection of BE and Barrett's neoplasia in tissue biopsies. 22 oesophageal tissue samples were collected from 19 patients. Contiguous frozen tissue sections were taken for pathology review and FTIR imaging. 45 mid-IR images were measured on an Agilent 620 FTIR microscope with an Agilent 670 spectrometer. Each image covering a 140 μm × 140 μm region was measured in 5 minutes, using a 1.1 μm2 pixel size and 64 scans per pixel. Principal component fed linear discriminant analysis was used to build classification models based on spectral differences, which were then tested using leave-one-sample-out cross validation. Key biochemical differences were identified by their spectral signatures: high glycogen content was seen in normal squamous (NSQ) tissue, high glycoprotein content was observed in glandular BE tissue, and high DNA content in dysplasia/adenocarcinoma samples. Classification of normal squamous samples versus ‘abnormal’ samples (any stage of Barrett's) was performed with 100% sensitivity and specificity. Neoplastic Barrett's (dysplasia or adenocarcinoma) was identified with 95.6% sensitivity and 86.4% specificity. Highly accurate pathology classification can be achieved with FTIR measurement of frozen tissue sections in a clinically applicable timeframe.
- This article is part of the themed collection: Optical Diagnosis
 Please wait while we load your content...
Please wait while we load your content...