Metabolomics as a tool to evaluate the toxicity of formulations containing amphotericin B, an antileishmanial drug†
Abstract
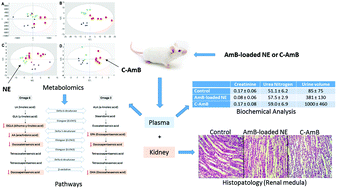
Amphotericin B (AmB) is a drug of choice against life-threatening systemic fungal infections and an alternative therapy for the treatment of all forms of leishmaniasis. It is known that AmB and its conventional formulation cause renal damage; however, the lipid formulations can reduce these effects. The aim of the present study was to identify metabolic changes in mice treated with two different AmB formulations, a nanoemulsion (NE) (lipid system carrier) loaded with AmB and the conventional formulation (C-AmB). For this purpose, metabolic fingerprinting represents a valuable strategy to monitor, in a non-targeted manner, the changes that are at the base of the toxicity mechanism of AmB. Plasma samples of BALB-c mice were collected after treatment with 3 alternate doses of AmB at 1 mg kg−1 administered intravenously and analysed with CE, LC and GC coupled to MS. Blood urea nitrogen (BUN) and plasma creatinine levels were also analysed. Kidney tissue specimens were collected and evaluated. It was not observed that there were any alterations in BUN and creatinine levels as well as in histopathological analysis. Approximately 30 metabolites were identified as potentially related to early C-AmB-induced nephrotoxicity. Disturbances in the arachidonic acid, glycerophospholipid, acylcarnitine and polyunsaturated fatty acid (PUFA) pathways were observed in C-AmB-treated mice. In the AmB-loaded NE group, it was observed that there were fewer metabolic changes, including changes in the plasma levels of cortisol and pyranose. The candidate biomarkers revealed in this study could be useful in the detection of the onset and severity of kidney injury induced by AmB formulations.

 Please wait while we load your content...
Please wait while we load your content...